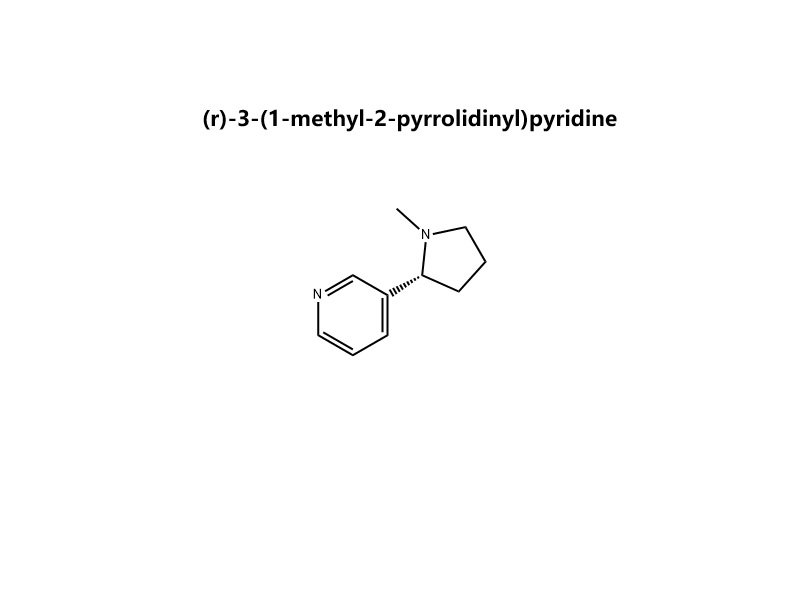
Nome IUPAC: (R)-3-[(2S)-1-Metilpirrolidin-2-il]piridina ou (R)-1-Metil-2-(3-piridil)pirrolidina
Nome comum: (R)-(−)-Nicotina; Nicotina Natural
Fórmula química: C₁₀H₁₄N₂
Peso molecular:162,23 g/mol
Rotação específica: [α]²⁰_D = aproximadamente -166° (puro)
Estrutura: Uma piridina (C₅H₄N) substituída na posição 3 com um grupo 1-metilpirrolidin-2-il com configuração (R).
Aparência: Líquido oleoso, transparente, de cor amarelo-pálido a acastanhado. Escurece quando exposto ao ar e à luz.
Ponto de ebulição: ~ 247 °C (decompõe-se)
Ponto de fusão: < -80°C (Líquido à temperatura ambiente)
Densidade: ~1,01 g/cm³ a 20°C
Pressão de vapor:~0,0425 mmHg a 20°C
Solubilidade: Miscível com água e a maioria dos solventes orgânicos (etanol, éter dietílico, clorofórmio) em todas as proporções a temperaturas abaixo de 60°C. Atua como uma base fraca (pKa do ácido conjugado: piridínio ~3,1, pirrolidínio ~7,9).
Estabilidade: Instável em condições alcalinas; volátil com vapor. Oxida-se e escurece após exposição prolongada ao ar. Os sais (ex.: dicloridrato, tartarato) são mais estáveis para armazenamento.
Farmacologia primária: A agonista potente dos receptores nicotínicos de acetilcolina (nAChRs)nos sistemas nervosos central e periférico. Essa atividade é a base de todos os seus efeitos fisiológicos.
Efeitos: Atua como estimulante em baixas doses, aumentando a frequência cardíaca, a pressão arterial e a liberação de neurotransmissores como dopamina (associada à recompensa e ao vício) e norepinefrina. Altas doses atuam como um agente bloqueador despolarizante nas junções neuromusculares, levando à toxicidade.
Toxicidade: Altamente tóxico (DL50 oral, rato: ~50 mg/kg). Os sintomas de intoxicação aguda incluem náuseas, vômitos, taquicardia, hipertensão, convulsões e insuficiência respiratória. Um conhecido teratógeno e substância vicianteDevido à sua rápida ação nos circuitos de recompensa do cérebro.
Metabolismo: Metabolizado principalmente no fígado pelo citocromo P450 2A6 (CYP2A6) em cotinina (principal metabólito) e outros compostos.
Biossíntese Natural: Na planta do tabaco (Nicotiana tabacum), é sintetizado nas raízes e transportado para as folhas. A via metabólica inicia-se com ácido aspártico e gliceraldeído-3-fosfato, passando por uma série de etapas que envolvem putrescina, N-metilputrescina e o intermediário crucial, o ácido nicotínico. O anel pirrolidínico deriva da ornitina/putrescina e o anel piridínico do ácido nicotínico (um derivado do aspartato). A enzima final, nicotina A sintase catalisa a reação de acoplamento, produzindo o enantiômero (R)-(−) naturalmente exclusivo.
Produção sintética: Pode ser produzido via síntese química ou extração e purificaçãoA partir de biomassa ou resíduos de tabaco, são utilizadas rotas sintéticas avançadas (síntese assimétrica, resolução enzimática) para produzir (R)-nicotina de alta pureza, evitando o enantiômero (S) indesejado, para aplicações farmacêuticas e de consumo avançado.

Produtos farmacêuticosTerapias para parar de fumar: O ingrediente ativo em Terapia de Reposição de Nicotina (TRN) Produtos (gomas de mascar, adesivos, pastilhas, inaladores) para aliviar os sintomas de abstinência.
Pesquisa Neurológica: Uma ferramenta padrão em pesquisa pré-clínica para estudar a função dos receptores nicotínicos de acetilcolina (nAChR), os mecanismos de dependência e potenciais terapias para a doença de Parkinson, declínio cognitivo e colite ulcerativa (onde os adesivos de nicotina demonstraram algum efeito).

AgroquímicosHistoricamente usado como um potente inseticida natural(atuando nos receptores nicotínicos de acetilcolina (nAChRs) dos insetos). Seu uso agora é altamente restrito ou proibido em muitos países devido à toxicidade para humanos.

Cigarros eletrônicos e líquidos para vaporizaçãoO principal agente viciante em produtos que contêm nicotina e-líquidosA mudança em direção à "nicotina sem tabaco" (TFN) geralmente se refere à (R)-nicotina produzida sinteticamente, comercializada por sua pureza e consistência.
Principais vantagens e benefícios
1. Especificidade e potência biológicas incomparáveis
Benefício: O O enantiômero (R) é a forma exclusiva que ocorre naturalmente.com estrutura tridimensional otimizada para ligação aos subtipos de nAChR de mamíferos. Isso garante efeitos precisos, reproduzíveis e biologicamente relevantesem sistemas experimentais.
Cenário de aplicação: Em pesquisa avançada em neurofarmacologiaAo estudar as vias de dependência ou a função cognitiva, o uso da (R)-nicotina elimina a variável de confusão do enantiômero (S) inativo, garantindo que os efeitos observados na liberação de dopamina ou na ativação neuronal sejam precisos e atribuíveis exclusivamente à forma ativa.
2. Fundamental para Pesquisa Validada e Padronização
Beneficiar:Serve como o padrão de referência definitivo para quantificar os níveis de nicotina e estudar seu metabolismo, toxicologia e farmacocinética.
Cenário de aplicação: Em toxicologia forense ou laboratórios de controle de qualidade farmacêuticaÉ utilizado para calibrar sistemas HPLC-MS/MS para a medição precisa de nicotina e seus metabólitos (como a cotinina) em amostras biológicas, garantindo resultados legal e medicamente defensáveis.
3. Essencial para o desenvolvimento moderno de produtos de nicotina
Beneficiar:Fornece um Ingrediente farmacêutico ativo (IFA) consistente e de alta pureza.Livre de impurezas específicas do tabaco (TSNAs) que podem surgir durante a extração, o que é crucial para os perfis de segurança.
Cenário de aplicação: No desenvolvimento de terapias de reposição de nicotina (TRN) de próxima geração ou sachês regulamentados sem tabacoA nicotina sintética (R) permite uma dosagem precisa, perfis de sabor mais puros e um argumento regulatório mais robusto em relação à pureza e caracterização do produto.
4. Possibilita estudos de relação estrutura-atividade (SAR)
Beneficiar:O enantiômero puro é o andaime fundamentalPara que químicos medicinais possam projetar e testar novos análogos para potenciais terapias direcionadas aos receptores nicotínicos de acetilcolina (nAChRs).
Cenário de aplicação: Em programas de descoberta de medicamentos para distúrbios neurológicos(por exemplo, Parkinson, esquizofrenia, dor), os pesquisadores modificam a estrutura da (R)-nicotina para criar novos compostos com atividade seletiva em subunidades de receptores, visando manter os benefícios cognitivos e, ao mesmo tempo, eliminar o potencial de dependência e os efeitos colaterais.
(R)-3-(1-Metil-2-pirrolidinil)piridina (Nicotina natural (R)-(−)-, CAS 25162-00-9) não é um produto químico básico, mas sim um Ferramenta bioquímica altamente especializada e ingrediente ativo de precisãoSua principal vantagem é a sua pureza enantiomérica, que está diretamente ligado ao seu eficácia biológica, validade da pesquisa e aceitabilidade regulatóriaÉ indispensável para Pesquisa neurológica de ponta, padronização analítica e desenvolvimento de produtos avançados e bem caracterizados para administração de nicotina.Para pesquisadores e desenvolvedores de produtos, obter nicotina (R) de alta pureza é um investimento em integridade de dados, consistência do produto e credibilidade científica, diferenciando seu trabalho de estudos ou produtos que dependem de misturas racêmicas menos definidas.
Perguntas frequentes (focadas no cliente)
Q1: Qual a diferença entre (R)-nicotina e nicotina racêmica?
A: O O enantiômero (R) é a forma biologicamente ativa que ocorre naturalmente. com alta afinidade pelos receptores nicotínicos de acetilcolina (nAChRs). A nicotina racêmica (uma mistura 50/50 das formas (R) e (S)) é frequentemente um subproduto da síntese química não estereosseletiva. O enantiômero (S) apresenta afinidade significativamente menor pelo receptor e farmacocinética diferente. Para pesquisa e aplicações de ponta, enantiômeros A pureza (>99% (R)) é crítica. Para uma resposta biológica precisa e um desempenho consistente do produto.
Q2: Quais são os requisitos críticos de manuseio e armazenamento?
UM: Extrema cautela é necessáriaDeve ser tratado em um capela de exaustãoCom EPI apropriado (luvas, óculos de proteção, jaleco). Armazene em local arejado. atmosfera inerte (Argônio/N₂) em um recipiente escuro e hermeticamente fechado at 2-8°CComprar e armazenar isso como um forma de sal estável (por exemplo, dicloridrato) é fortemente recomendado para maior segurança e prazo de validade. Consulte sempre a Ficha de Dados de Segurança (FDS).
Q3: Quais são os graus de pureza disponíveis e qual devo especificar para minha aplicação?
A:
Grau técnico (aproximadamente 95%):Para aplicações industriais não críticas (ex.: pesquisa de inseticidas).
Grau farmacêutico (pureza química >99%): Obrigatório para o desenvolvimento de produtos NRT.
Alto grau de pureza enantiomérica (>99,5% (R)): Essencial para Pesquisa avançada em neurofarmacologia e TFN sintético para líquidos de vaporização.Onde a interação precisa com o receptor e o perfil de sabor são fundamentais. Sempre especifique. pureza química, excesso enantiomérico (ee) e níveis de solvente residual.
Q4: Qual é o panorama regulatório para a compra e utilização deste composto?
A: É fortemente regulamentadocomo substância controlada/precursora na maioria das jurisdições.
EUA: Regulamentado pela FDA e pela DEA. As vendas são frequentemente restritas a instituições de pesquisa qualificadas e fabricantes licenciados.
UE: Sujeitos a rigorosos controles de acordo com as regulamentações de segurança química e do consumidor (por exemplo, REACH, CLP). Para produtos de vaporização, a Diretiva de Produtos de Tabaco (TPD) estabelece limites de concentração.
Conformidade: Os compradores devem fornecer documentação de uso finale comprovação de conformidade com as regulamentações locais. Para uso comercial em produtos de consumo (cigarros eletrônicos), são necessários dossiês toxicológicos abrangentes e autorização de comercialização.
Q5: Pode ser usado como padrão em testes analíticos?
UM: Sim, é o padrão de referência primário.Para quantificar a nicotina em amostras de tabaco, farmacêuticas e toxicológicas por meio de HPLC, GC-MS ou LC-MS. Para isso, você deve obter um Material de Referência Padrão Certificado (CRM)De um fornecedor qualificado, com certificado de análise que detalhe a pureza exata e a rastreabilidade.
Q6: Quais são as principais considerações logísticas e de fornecimento?
A: Devido à sua classe de perigo (6.1, tóxico), transporte especializado de mercadorias perigosas (HAZMAT)É obrigatório, aumentando o custo e a complexidade. Os prazos de entrega podem ser longos. Fonte: fornecedores de ingredientes químicos finos ou farmacêuticos de boa reputação e estabelecidos Com experiência comprovada no manuseio de alcaloides. Desconfie de discrepâncias nos preços, pois preços muito baixos podem indicar misturas racêmicas, baixa pureza ou fornecimento irregular.


 Português
Português English
English Español
Español Português
Português




![4-Nitrophenyl 3-(Benzo[d][1,3]dioxol-5-yl)-2-methylpropanoate](https://pt.qixiolivetol.com/storage/uploads/images/202604/17/1776427287_qZy87IjHSQ.jpg)

 CONTATE-NOS
CONTATE-NOS






 IPv6 SUPORTADO POR REDE
|
IPv6 SUPORTADO POR REDE
|

